Axonal Modeling
Development of mathematical models relating white matter microstructure to axonal signal transmission delays between different brain areas.
The white matter of the brain contains axons connecting distant brain areas, many of which are ensheathed in a myelin layer that increases transmission speed of neuronal signals. Although axonal signal transmission is fast, it still accounts for significant delays, which are critical for shaping cortical dynamics and synchronisation between brain areas. In addition to diffusion tractography, which is mainly sensitive to the trajectories of fiber tracts, advanced MRI techniques are able to reveal further aspects of white matter microstructure of non-invasively. This knowledge may be used to describe axonal transmission delays and thereby extend and refine mathematical models describing neuronal activity at the whole brain level.
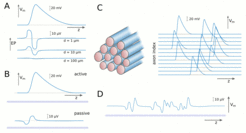
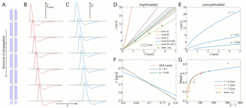
This project aims to establish the relationship of the velocity of axonal signal transmission with the structural parameters of white matter using various mathematical models. Of particular interest are the sensitivity of axonal transmission speed to structural and functional parameters, but also the possible effects of extracellular field potentials generated by neuronal signals on axonal signal transmission. In the first part of this project, we use mathematical models that describe the propagation of action potentials (so-called spikes) in single axonal fibres. A particular focus is on spike-diffuse-spike models, which model the action potential generation and propagation by threshold activated currents. In these models, the propagation of an action potential along a myelinated axon (Figure 1A) is driven by the shape of these currents (Figure 1B), which can be measured experimentally. The time course and the activation thresholds of these currents determine the shape of the temporal profile of an action potential (Figure 1C). With such models, one can obtain semi-analytical solutions for the spike velocity, which allows for large parameter sweeps. Microstructural parameters that can be possibly accessed non-invasively are the fibre diameters and the g-ratio, which is the ratio between the inner diameter (axon) and outer diameter (axon + myelin) of a myelinated axon. Our modelling work confirms some previously published results for the relationship between spike velocity and axon diameter in both myelinated and unmyelinated axons (Figure 1D and Figure 1E), but also reveals that the sensitivity to the g-ratio is stronger than previously thought (Figure 1F). The latter particularly depends on the ratio between internode and node length (Figure 1G).
The second part of the project is concerned with the generation of extracellular potentials by propagating spikes, and how these extracellular potentials affect their propagation velocity. Extracellular potentials can be computed from the currents passing along an axon as a result of spike generation. The extracellular potentials generated by a single spike (Figure 2A) and the resulting perturbations of resting potentials in nearby axons (Figure 2B) are usually quite weak (about 10 microvolts). However, if spike volleys containing a large number of spikes propagate along an axonal fibre bundle (Figure 2C), then the resulting extracellular potentials show a cumulative effect (Figure 2D). In macroscopic fibre bundles the extracellular potentials can reach several millivolts, and as a consequence, axons experience a significant modulation of their firing thresholds, which in turn affects their propagation velocities. We develop mathematical models that can efficiently describe this effect in axon bundles containing a large number of axons.